Calcium compounds like Calcium Carbide (CaC2) are the strongest deoxidizers in Secondary Metallurgy. Reaction model for CaC2-Deoxidation:
Calcium compounds like Calcium Carbide (CaC2) are the strongest deoxidizers in Secondary Metallurgy.
ALMAMET is your reliable partner for supplying Calcium Carbide for steel deoxidation, pure as well as pre-mixed with slag conditioners. For safe and precise usage ALMAMET offers automatic dosing systems to ensure a safe handling.
Calcium compounds like Calcium Carbide (CaC2) are the strongest deoxidizers in Secondary Metallurgy. Reaction model for CaC2-Deoxidation:
The deoxidation with CaC2 is independently possible in several stages of steelmaking process:


Using CaC2 as deoxidizer in Secondary Metallurgy leads to many benefits in the whole steelmaking process:
ALMAMET offers technical Calcium Carbide in different grain sizes and tailor-made packaging.
Carbide-based pellets for slag conditioning during LF-treatment can be produced containing additives for enhanced properties.
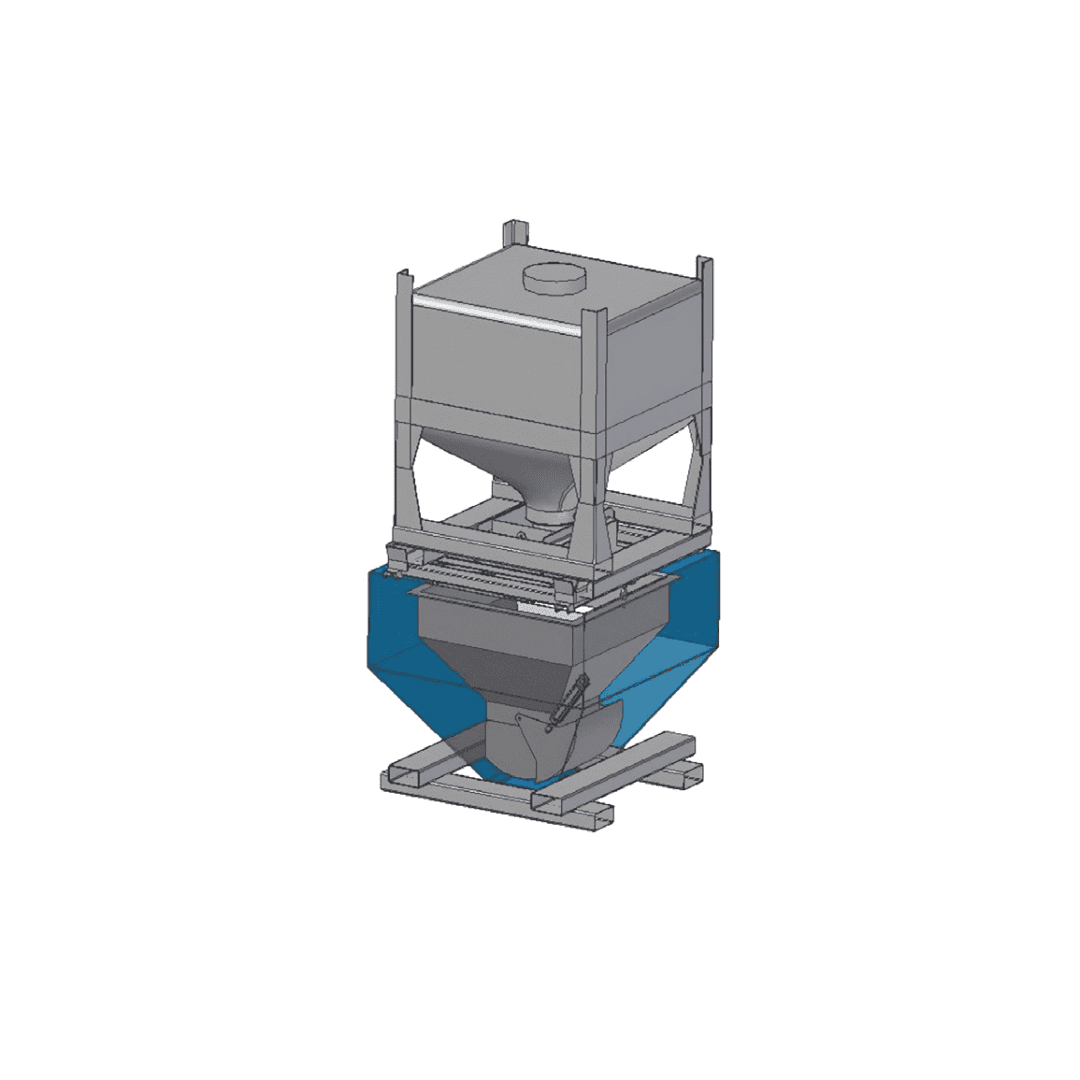
ALMAMET engineers and supplies tailor-made dosing systems that enable the safe addition of precise amounts of lumpy Calcium Carbide during tapping at BOF or EAF operations.
The dosing systems are engineered to precisely control both the quantity and the dosing rate of Calcium Carbide, ensuring maximum safety while enhancing steel quality and reducing operational costs.